1.1.1.184: carbonyl reductase (NADPH)
This is an abbreviated version!
For detailed information about carbonyl reductase (NADPH), go to the full flat file.
Reaction
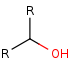 R-CHOH-R' +
R-CHOH-R' +
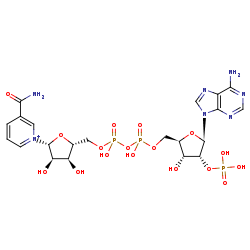 NADP+=
NADP+=
 R-CO-R' +
R-CO-R' +
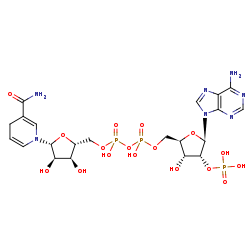 NADPH +
NADPH +
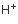 H+
H+
Synonyms
(R) specific carbonyl reductase, (S)-specific carbonyl reductase, 15-hydroxyprostaglandin dehydrogenase [NADP+], 2,5-diketo-D-gluconic acid reductase, Adipocyte P27 protein, aldehyde reductase 1, aldehyde reductase I, aldo-keto reductase, ALR3, AP27, carbonyl reductase, carbonyl reductase (NADPH), carbonyl reductase 1, carbonyl reductase 3, carbonyl reductase S1, CBR, CBR 1, CBR 3, CBR1, CBR3, CHCR, CHCR1, CHCR2, CHCR3, CR, CR125, crc1, CSCR1, Gox0644, Gox1615, hCBR1, ketoreductase, KLCR1, KR, LCR, LOC415661, LOC610164, microsomal carbonyl reductase, More, NADP+-dependent ADH, NADPH-carbonyl reductase, NADPH-dependent carbonyl reductase, NADPH-dependent carbonyl reductase S1, NCCR, nonspecific NADPH-dependent carbonyl reductase, peroxisomal-type carbonyl reductase, PHCR, prostaglandin 9-ketoreductase, Prostaglandin-E2 9-reductase, PTCR, R-specific carbonyl reductase, reductase S1, reductase, carbonyl, RLCR, SCR, SCR9, SDR21C1, secondary-alcohol: NADP+-oxidoreductase, short-chain (S)-1-phenyl-1,2-ethanediol dehydrogenase, short-chain carbonyl reductase, sniffer, SRED, SSCR, SyS1, tetrameric carbonyl reductase, Tm1743, Tm_1743, xenobiotic carbonyl reductase, xenobiotic ketone reductase, YGL039w1, YGL039w2, YtbE, YueD
ECTree
KCat KM Value
KCat KM Value on EC 1.1.1.184 - carbonyl reductase (NADPH)
Please wait a moment until all data is loaded. This message will disappear when all data is loaded.
Please wait a moment until the data is sorted. This message will disappear when the data is sorted.
80
(R)-2-butanol
-
pH 7.5, 37°C
27
(S)-2-butanol
-
pH 7.5, 37°C
50 - 56.7
1,2-naphthoquinone
8.7 - 73.3
1,4-Naphthoquinone
4 - 5.75
1-(3-methyl-1,4-dioxidoquinoxalin-2-yl)ethanone
25 - 26
1-(3-methyl-4-oxidoquinoxalin-2-yl)ethanone
1.4
2,3-Butanediol
-
pH 7.5, 37°C
25 - 31.7
2,3-Hexanedione
0.99 - 6.4
2,3-Pentanedione
42
2-butanol
-
pH 7.5, 37°C
9
2-butanone
-
pH 7.5, 37°C
1.3
2-Pentanone
-
pH 7.5, 37°C
81
2-propanol
-
pH 7.5, 37°C
1281
3-glutathionyl-hexanal
pH 8.4, 25°C
4615
3-glutathionyl-nonanal
pH 8.4, 25°C
885
3-glutathionyl-propanal
pH 8.4, 25°C
0.17 - 0.25
4-(methylnitrosamino)-1-(3-pyridyl)-1-butanone
4.8 - 15.7
4-benzoylpyridine
113 - 2828
9,10-phenanthrenequinone
11
acetoin
-
pH 7.5, 37°C
13
cyclohexanone
-
pH 7.5, 37°C
23
Cyclopentanol
-
pH 7.5, 37°C
19
Cyclopentanone
-
pH 7.5, 37°C
203
diacetyl
-
pH 7.5, 37°C
11
methylglyoxal
pH 7.0, 25°C
11600
NADP+
-
pH 7.5, 37°C
8.2
Phenylglyoxal
pH 7.0, 25°C
321
propanone
-
pH 7.5, 37°C
1.72 - 93.45
S-nitrosoglutathione
50
1,2-naphthoquinone

wild-type, pH 7.4, 25°C
50
1,2-naphthoquinone
wild-type, pH 7.4, 37°C
56.7
1,2-naphthoquinone
after treatment with S-nitrosoglutathione, pH 7.4, 25°C
56.7
1,2-naphthoquinone
enzyme modified by S-nitrosoglutathione, pH 7.4, 37°C
8.7
1,4-Naphthoquinone

after treatment with S-nitrosoglutathione, pH 7.4, 25°C
8.7
1,4-Naphthoquinone
enzyme modified by S-nitrosoglutathione, pH 7.4, 37°C
73.3
1,4-Naphthoquinone
wild-type, pH 7.4, 25°C
73.3
1,4-Naphthoquinone
wild-type, pH 7.4, 37°C
4
1-(3-methyl-1,4-dioxidoquinoxalin-2-yl)ethanone

pH 7.4, 37°C
5.75
1-(3-methyl-1,4-dioxidoquinoxalin-2-yl)ethanone
pH 7.4, 37°C
25
1-(3-methyl-4-oxidoquinoxalin-2-yl)ethanone

pH 7.4, 37°C
26
1-(3-methyl-4-oxidoquinoxalin-2-yl)ethanone
pH 7.4, 37°C
25
2,3-Hexanedione

after treatment with S-nitrosoglutathione, pH 7.4, 25°C
25
2,3-Hexanedione
enzyme modified by S-nitrosoglutathione, pH 7.4, 37°C
31.6
2,3-Hexanedione
wild-type, pH 7.4, 25°C
31.7
2,3-Hexanedione
wild-type, pH 7.4, 37°C
0.99
2,3-Pentanedione

pH 7.0, 25°C
6.4
2,3-Pentanedione
pH 7.0, 25°C
0.17
4-(methylnitrosamino)-1-(3-pyridyl)-1-butanone

enzyme modified by S-nitrosoglutathione, pH 7.4, 37°C
0.25
4-(methylnitrosamino)-1-(3-pyridyl)-1-butanone
wild-type, pH 7.4, 37°C
4.8
4-benzoylpyridine

wild-type, pH 7.4, 25°C
4.8
4-benzoylpyridine
wild-type, pH 7.4, 37°C
15.7
4-benzoylpyridine
after treatment with S-nitrosoglutathione, pH 7.4, 25°C
15.7
4-benzoylpyridine
enzyme modified by S-nitrosoglutathione, pH 7.4, 37°C
113
9,10-phenanthrenequinone

wild-type, pH 7.4, 25°C
113
9,10-phenanthrenequinone
wild-type, pH 7.4, 37°C
148.3
9,10-phenanthrenequinone
wild-type, pH 7.4, temperature not specified in the publication
533
9,10-phenanthrenequinone
after treatment with S-nitrosoglutathione, pH 7.4, 25°C
533
9,10-phenanthrenequinone
enzyme modified by S-nitrosoglutathione, pH 7.4, 37°C
2828
9,10-phenanthrenequinone
mutant C227S, pH 7.4, temperature not specified in the publication
11.2
daunorubicin

enzyme modified by S-nitrosoglutathione, pH 7.4, 37°C
18.3
daunorubicin
wild-type, pH 7.4, 37°C
0.035
doxorubicin

isoform Cbr3, at pH 7.0 and 37°C
0.12
doxorubicin
isoform Cbr1, at pH 7.0 and 37°C
18.3
isatin

after treatment with S-nitrosoglutathione, pH 7.4, 25°C
18.3
isatin
enzyme modified by S-nitrosoglutathione, pH 7.4, 37°C
21.7
isatin
wild-type, pH 7.4, 25°C
21.7
isatin
wild-type, pH 7.4, 37°C
7.5
menadione

pH 7.4, 37°C
21.7
menadione
after treatment with S-nitrosoglutathione, pH 7.4, 25°C
21.7
menadione
enzyme modified by S-nitrosoglutathione, pH 7.4, 37°C
38.3
menadione
wild-type, pH 7.4, 25°C
38.3
menadione
wild-type, pH 7.4, 37°C
54.5
menadione
pH 7.4, 37°C
0.0013
NADPH

isoform Cbr3, at pH 7.0 and 37°C
0.36
NADPH
isoform Cbr1, at pH 7.0 and 37°C
5200
NADPH
-
pH 7.5, 37°C
11.3
oracin

enzyme modified by S-nitrosoglutathione, pH 7.4, 37°C
12.2
oracin
wild-type, pH 7.4, 37°C
1.72
S-nitrosoglutathione

mutant D236A/K238P/D239K/S240A/I241T/R242K/T243S/V244P, pH 7.4, temperature not specified in the publication
2.5
S-nitrosoglutathione
mutant C227S, pH 7.4, temperature not specified in the publication
9.93
S-nitrosoglutathione
mutant P230W/D236A/K238P/D239K/S240A/I241T/R242K/T243S/V244PpH 7.4, temperature not specified in the publication
24.58
S-nitrosoglutathione
mutant Q142M/C143S/P230W/D236A/K238P/D239K/S240A/I241T/R242K/T243S/V244P/H270S, pH 7.4, temperature not specified in the publication
93.45
S-nitrosoglutathione
wild-type, pH 7.4, temperature not specified in the publication



 results (
results ( results (
results ( top
top






